1. State differences between acids and bases.
Ans.
Q.2.Ammonia is found in many household products, such as window cleaners. It turns red litmus blue. What is its nature?
Ans.Ammonia has basic nature.
Q.3.Name the source from which litmus solution is obtained. What is the use of this solution?
Ans.Litmus solution is extracted from lichens. It is used to determine whether the given solution is acidic or basic.
Q.4.Is the distilled water acidic/basic/neutral? How would you verify it?
Ans.Distilled water will be neutral. We can verify it by showing that neither blue nor red litmus paper changes its colour when dipped in it.
Q.5.Describe the process of neutralisation with the help of an example.
Ans.The reaction between an acid and a base is known as neutralisation. Salt and water are produced in this process with the evolution of heat.
Antacids like milk of magnesia (magnesium hydroxide), baking soda, etc. which contain a base are used for reducing acidity in stomach when excessive acid released by glands.
Q.6.Mark ‘T’ if the statement is true and ‘F’ if it is false:
(i) Nitric acid turns red litmus blue. (T/F)
(ii) Sodium hydroxide turns blue litmus red. {T/F)
(iii) Sodium hydroxide and hydrochloric acid neutralise each other and form salt and water. (T/F)
(id) Indicator is a substance which shows different colours in acidic and basic solutions. . (T/F)
(v) Tooth decay is caused by the presence of a base. (T/F)
Ans.(1) F (ii) F (iii) T (iv) T (V) F
Q.7. Dorji has a few bottles of soft drink in his restaurant. But, unfortunately, these are
not labelled. He has to serve the drinks on the demand of customers. One customer
wants acidic drink, another wants basic and third one wants neutral drink. How
will Dorji decide which drink is to be served to whom?
Ans.Dorji can decide with the help of litmus paper:
(i) The drink which would turn a red litmus blue would be basic.
(ii) If the drink turns a blue litmus to red would be acidic.
(iii) The drink which would not affect both red and blue litmus would be neutral.
Q.8.Explain why:
(a) An antacid tablet is taken when you suffer from acidity.
(b) Calamine solution is applied on the skin when an ant bites.
(c) Factory waste is neutralised before disposing it into the water bodies.
Ans.(a) We take an antacid such as milk of magnesia to neutralises the excessive acid released in stomach.
(b) Ant injects an acidic liquid (Formic acid) into the skin on biting which causes inflammation, to the skin. The effect of the acid can be neutralised by rubbing. Calamine solution which contains zinc carbonate which is very weak base and causes no harm to the skin.
(c) The wastes of factories contain acids. If acids are disposed off in the water body, the acids will harm the organisms. So factory wastes are neutralised by adding basic substances.
Q.9. Three liquids are given to you. One is hydrochloric acid, another is sodium hydroxide and third is a sugar solution. How will you identify them? You have only turmeric indicator.
Ans.Name of the substances Effect on turmeric indicator
1. Hydrochloric acid Yellow to blue
2. Sodium hydroxide Yellow to red
3. Sugar solution No change
Q.10. Blue litmus paper is dipped in a solution. It remains blue. What is the nature of the solution? Explain.
Ans. (i) It can be identified on the basis of the following observations : Bases change the colour of litmus paper to blue. As the colour of blue litmus paper is not affected, the solution must be basic.
(ii) If the solution is neutral, even then colour of litmus will not change.
Q. 11. Consider the following statements:
(a) Both acids and bases change colour of all indicators.
(b) If an indicator gives a colour change with an acid, it does not give a change with a base.
(c) If an indicator changes colour with a base, it does not change colour with an acid.
(d) Change of colour in an acid and a base depends on the type of the indicator. Which of these statements are correct?
(i) All four (ii) (a) and (d) (iii) (b) and (c) (iv) only (d)
Ans. (ii) (a) and (d)
Acids, Bases and Salts Class 7 Extra Questions Science Chapter 5
Acids, Bases and Salts Class 7 Science Extra Questions Very Short Answer Type Questions
Question 1.
What is the test for acids and bases using litmus paper?
Answer:
Acids turn blue litmus paper red while bases turn red litmus paper blue.
Question 2.
What name is given to those substances which do not change the colour of either red or blue litmus paper?
Answer:
The solutions or substances which do not change the colour of either red or blue litmus paper are neutral.
Question 3.
Write the name of two indicators which occur naturally.
Answer:
- Turmeric
- China rose
Question 4.
Name the acid present in lemon juice and tomato.
Answer:
Citric acid in lemon juice and oxalic acid in tomato.
Question 5.
Name a substance which is used to cure the discomfort caused by indigestion due to over eating.
Answer:
Milk of magnesia which contains magnesium hydroxide (base) is used to neutralise the effect of excessive acid in stomach.
Question 6.
Why does a turmeric stain on white shirt is turned to red when it is washed with soap? [HOTS]
Answer:
Turmeric stain on white shirt is turned to red when it is washed with soap because the soap solution is basic in nature.
Question 8.
Is lime water an acidic or basic solution?
Answer:
Lime water (calcium hydroxide) is a basic solution.
Question 9.
Name the reaction when an acid is mixed with base.
Answer:
Neutralisation reaction.
Question 10.
Which acid is injected into our body by an ant sting that causes pain? [HOTS]
Answer:
The acid is formic acid which is injected into the skin of a person when an ant bites.
Question 11.
Name the salt formed when hydrochloric acid reacts with sodium hydroxide solution (a base).
Answer:
When hydrochloric acid reacts with sodium hydroxide solution, then sodium chloride salt is formed.
Question 12.
Write two basic substances which we use in our daily life.
Answer:
- Baking soda
- Soap or detergent
Question 13.
Name one acid which is responsible for acid rain.
Answer:
Carbonic acid.
Question 14.
Name two gaseous pollutants which are responsible for acid rain.
Answer:
Two gaseous pollutants are nitrogen dioxide (NO2) and sulphur dioxide (SO2).
Question 15.
(a) Name one indicator which turns red on adding an acid.
(b) Name one indicator which turns red on adding a base.
Answer:
(a) Litmus (blue litmus paper)
(b) Turmeric
Question 16.
Form a sentence using the following words: baking soda, ant bite, moist, effect, neutralised, rubbing [NCERT Exemplar; HOTS]
Answer:
The effect of an arft bite can be neutralised by rubbing moist baking soda.
Acids, Bases and Salts Class 7 Science Extra Questions Short Answer Type Questions
Question 1.
Write the characteristics of acids by which we can identify acids.
Answer:
Characteristics of acids are
- They are sour in taste.
- They turn blue litmus paper/solution to red.
- Dilution of acid in water is an exothermic reaction, i.e. heat energy is evolved.
Question 2.
Look at the figure which shows solutions taken in test tubes A, B, C and D. What colour is expected, when a piece of red litmus paper is dropped in each test tube? Nature of the solutions is given in the table for your help.

| Test tube | Nature of solution | Change in colour of red litmus |
| A | Neutral | |
| B | Basic | |
| C | Acidic | |
| D | Neutral |
[NCERT Exemplar]
Answer:
Nature of solution is given below:
|
Test tube |
Nature of solution | Change in colour of red litmus |
| A | Neutral | No change |
| B | Basic | Turn blue |
| C | Acidic | No change |
| D | Neutral | No change |
Question 3.
While playing in a park, a child was stung by a wasp. Some elders suggested applying paste of baking soda and others lemon juice as remedy. Which remedy do you think is appropriate and why? [NCERT Exemplar; HOTS]
Answer:
Wasp sting inject a liquid in the skin which is acidic in nature. Hence, baking soda is the appropriate remedy as it is basic in nature and neutralises the acid.
Question 4.
Which of the following are acidic and which are basic?
Lime water, Vinegar, Toothpaste, Stomach juices, Lemon juice, Baking soda solution, Milk of magnesia, Ammonia solution.
Answer:
| Acidic in nature | Basic in nature |
| Vinegar | Lime water |
| Stomach juices | Toothpaste |
| Lemon juice | Baking soda solution |
| Milk of magnesia | |
| Ammonia solution |
Question 5.
A small amount of hydrochloric acid is always produced in the stomach. Is it useful or harmful for us? If excess of acid is produced in the stomach, what should we do?
Answer:
A small amount of hydrochloric acid produced in the stomach is useful as it can kill the harmful bacteria that may enter into the stomach along with the food.
However, if excess of acid is produced, there is a burning sensation in the stomach. We should take milk of magnesia as an antacid medicine to neutralise the excess acid.
Question 6.
Write the effect of China rose petals on acidic and basic solutions.
Answer:
China rose petals when added to warm water, form a light pink coloured solution which may be used as an indicator. This indicator turns acidic solution to magenta (deep pink) and basic solution to green.
Question 7.
To test the presence of an acid in any substance, what methods can be used?
Answer:
Acid can be tested in any substance by the following methods
- Take a drop of the dilute solution of the substance on the tips of your tongue. If tastes sour, it is acidic.
- Dip a blue litmus paper in the solution. If the colour of the litmus paper turns red, the solution is acidic.
Question 8.
Why are sodium bicarbonate and lemon juice used during indigestion?
Answer:
Sodium bicarbonate neutralises the acidity in the stomach. Hence, it is used during indigestion. Lemon contains acid. It reacts with undigested food and suffers it.
Question 9.
After carrying out the neutralisation reaction, the test tube immediately found to be somewhat hot. Explain why.
Answer:
In neutralisation reaction, heat is always produced or evolved. The evolved heat raises the temperature of the reaction mixture. Therefore, if we touch the test tube immediately after the neutralisation reaction, it is found to be hot.
Acid + Base → Salt + Water (heat is evolved)
Question 10.
Name three acids used in the laboratory.
Answer:
Acids which are mostly used in laboratory as below
- Hydrochloric acid (HCl)
- Sulphuric acid (H2SO4)
- Nitric acid (HNO3)
Question 11.
Paheli is suffering from indigestion due to acidity. Is it advisable to give her orange juice in this situation and why? |NCERT Exemplar; HOTS]
Answer:
No, because orange juice is acidic in nature. Excess of acid in the stomach causes indigestion. We take an antacid such as milk of magnesia which contains magnesium hydroxide.
Question 12.
Explain two neutralisation reactions related to daily life situations.
Answer:
(i) Ant bite When an ant bite injects the acidic liquid (formic acid) into the skin, the effect of the acid can be neutralised by rubbing moist baking soda (sodium hydrogen carbonate) or calamine solution, which contains zinc carbonate.
(ii) Indigestion Our stomach contains hydrochloric acid. It helps us to digest food but too much of acid in the stomach causes indigestion. Sometimes, indigestion is painful. To relieve indigestion, we take an antacid such as milk of magnesia which contains magnesium hydroxide.
It neutralises the effect of excessive acid.
Question 13.
How lime water is prepared in the laboratory?
Answer:
To prepare lime water, dissolve some lime (chuna) in water in a bottle. Stir the solution and keep it for sometime. Pour a little more from the top. This is lime water.
Question 14.
Name three types of salts. Give one example of each type of salt.
Answer:
Type of salts
- Neutral salts e.g. sodium chloride (NaCl)
- Acidic salts e.g. Ammonium chloride (NH4Cl)
- Basic salts e.g. sodium hydrogen carbonate (NaHCO3)
Question 15.
Paheli observed that most of the fish in the pond of her village were gradually dying. She also observed that the wastes of a factory in their village are flowing into the pond which probably caused the fish to die.
(a) Explain why the fish were dying?
(b) If the factory waste is acidic in nature, how can it be neutralised? [NCERT Exemplar; HOTS]
Answer:
(a) If the wastes of a factory flow into waterbodies, it can cause a threat to the lives of sea creatures and to anybody who drink the water. Since, factory wastes may contain acids or bases and it can kill the fish.
(b) If the factory waste is acidic in nature, it can be neutralised by adding basic substances.
Question 16.
What is a salt? Name any salt and give their formulae.
Answer:
A substance formed by the neutralisation of an acid with a base is called salt. Salt may be acidic, basic or neutral in nature.
For example,
Hydrochloric acid (HCI) +
(Acid)
Sodium hydroxide (NaOH) →
(Base)
Sodium chloride (NaCI) + Water (Salt)
(Salt)
Question 17.
Look at the given reaction.
Hydrochloric acid + Sodium hydroxide (base) → Sodium chloride (salt) + Water
Sodium chloride formed in this reaction remains in solution form. Can we get solid sodium chloride from this solution? Suggest a method (if any). [NCERT Exemplar]
Answer:
We can get solid sodium chloride by evaporation method. Evaporation is the process by which water changes from a liquid to a gas or vapour.
Rate of evaporation increases with temperature.
Question 18.
Name three bases used in the laboratory with their formulae.
Answer:
Bases which are mostly used in laboratory as below
- Sodium hydroxide (NaOH)
- Calcium hydroxide [Ca(OH)2]
- Ammonium hydroxide (NH4OH)
Acids, Bases and Salts Class 7 Science Extra Questions Long Answer Type Questions
Question 1.
A farmer was unhappy because of his low crop yield. He discussed the problem with an agricultural scientist and realised that the soil of his field was either too acidic or too basic. What remedy would you suggest the farmer to neutralise the soil? [HOTS]
Answer:
The reason for low crop yield in farmer’s field was that the soil at a place is either too acidic or too basic. The soil may be acidic or basic naturally. The excessive use of fertilisers in the fields also makes the soil too acidic. When the soil is too acidic, it is treated with base such as quicklime (calcium oxide) or slaked lime (calcium hydroxide) which neutralises the excess acid present in the soil and reduces its acidic nature. If the soil is too basic, then decaying organic matter (called manure or compost) is added it. The decaying organic matter releases acid which neutralises the excess bases present in the soil and reduces its basic nature.
Question 2.
You are provided with three test tubes A, Sand Cas shown in figure with different liquids. What will you observe when you put
(a) a piece of blue litmus paper in each test tube?
(b) a piece of red litmus paper in each test tube?
(c) a few drops of phenolphthalein solution to each test tube?
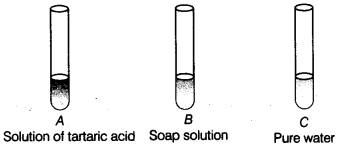
Answer:
| Test tube | Effect on blue litmus paper | Effect on red litmus paper | Effect on phenolphthalein solution |
| A | Turns red | Remains red | Colourless |
| B | Remains blue | Turns blue | Pink colour |
| C | Remains blue | Remains red | Colourless |
Question 3.
Boojho, Paheli and their friend Golu were provided with a test, each containing China rose solution which was pink in colour. Boojho added 2 drops of solution ‘A’ in his test tube and got dark pink colour. Paheli added 2 drops of solution ‘S’to her test tube and got green colour. Golu added 2 drops of solution ‘C but could not get any change in colour. Suggest the possible cause for the variation in their results. [HOTS]
Answer:
China rose (gudhal) is an acid-base indicator.
China rose solution in different medium shows following changes:
China rose + acid → Give dark pink colour, hence A is an acidic solution.
China rose + base → give green colour, hence 6 is a basic solution.
China rose + neutral → No change in colour, hence C is a neutral solution.
Question 4.
Nitesh was playing with his friends in the garden. Suddenly, Nitesh was stung by a honeybee and was in great pain. Immediately, his friends call his mother. She applied baking soda solution on the affected area and then took him to the doctor.
Read the above passage and answer the following questions:
(a) What could be the reason for this burning pain?
(b) Why did his mother applied baking soda solution on the affected area?
(c) What values are shown by Nitesh’s friends? [Value Based Question]
Answer:
(a) The reason for this burning pain is honeybee sting which causes pain and irritation. This is due to the acidic liquid (formic acid) injected into the skin by the honeybee.
(b) The effect of acid can be neutralised by rubbing the affected area by baking soda solution which is a mild base.
(c) Nitesh’s friends are caring , supportive and helpful.
Question 5.
One day Rahul’s mother after taking meal felt pain and irritation in her stomach. His father was out of station. Rahul was an intelligent boy. He remembered his teacher’s statement and gave his mother some baking soda solution Which gave her a relief from pain and irritation of stomach.
Read the above passage and answer the following questions.
(a) Which information given by Rahul’s teacher that helped him to select the baking soda as remedy?
(b) Why he selects baking soda as a cure?
(c) What values are shown by Rahul? [Value Based Question]
Answer:
(a) Rahul knows that our stomach contains hydrochloric acid. It helps us to digest food. But, too much acid in the stomach causes indigestion. Sometimes, indigestion is painful and some mild base should be taken to relief from this pain. So, this information given by his teacher helped him.
(b) He gave her mother baking soda solution which was available in the kitchen easily. He selects baking soda because it is a mild base and neutralises the excess acid in the stomach and it will give relief from pain.
(c) Rahul is very intelligent and caring boy.
Acids, Bases and Salts Class 7 Science Extra Questions Miscellaneous Questions
Multiple Choice Questions
Question 1.
The correct way of making a solution of acid in water is to [NCERT Exemplar]
(a) pdd water to acid
(b) add acid to water
(c) mix acid and water simultaneously
(d) add water to acid in a shallow container
Answer:
(b) add acid to water
Question 2.
Products of a neutralisation reaction are always
(a) an acid and a base
(b) an acid and a salt
(c) a salt and water
(d) a salt and a base
Answer:
(c) a salt and water
Question 3.
Turmeric is a natural indicator. On adding its paste to acid and base separately, which colours would be observed?
(a) Yellow in both acid and base
(b) Yellow in acid and red in base
(c) Pink in acid and yellow in base
(d) Red in acid and blue in base
Answer:
(b) Yellow in acid and red in base
Question 4.
Phenolphthalein is a synthetic indicator and its colours in acidic and basic solutions respectively are [NCERT Exemplar]
(a) red and blue
(b) blue and red
(c) pink and colourless
(d) colourless and pink
Answer:
(d) colourless and pink
Question 5.
When the soil is too basic, plants do not grow well in it. To improve its quality, what must be added to the soil? [NCERT Exemplar]
(a) Organic matter
(b) Quicklime
(c) Slaked lime
(d) Calamine solution
Answer:
(a) Organic matter
Question 6.
Neutralisation reaction is a [NCERT Exemplar]
(a) physical and reversible change
(b) physical change that cannot be reversed
(c) chemical and reversible change
(d) chemical change that cannot be reversed
Answer:
(d) chemical change that cannot be reversed
Question 7.
A solution changes the colour of turmeric indicator from yellow to red. The solution is
(a) basic
(b) acidic
(c) neutral
(d) either neutral or acidic
Answer:
(a) basic
Question 8.
Which of the following sets of substances contains acids?
(a) Grapes, lime water
(b) Vinegar, soap
(c) Curd, milk of magnesia
(d) Curd, vinegar
Answer:
(d) Curd, vinegar
Question 9.
Which of the following is an acid-base indicator? [NCERT Exemplar]
(a) Vinegar
(b) Lime water
(c) Turmeric
(d) Baking soda
Answer:
(c) Turmeric
Fill in the Blanks
1. Lemon juice and vinegar taste ……………… because they contain ……………… . [NCERT Exemplar]
2. Turmeric and litmus are ……………… acid-base indicators.
3. Phenolphthalein gives ……………… colour with lime water.
4. When an acidic solution is mixed with a basic solution, they ……………… each other forming ……………… and water. [NCERT Exemplar]
5. The acidic or basic nature of a substance is tested by using an ……………… .
6. The substances which show different colours in acidic, basic and neutral solutions are called ………………
7. Acid + Base → ……………… + Water
8. When an acid mixed with a base, a ……………… reaction takes place.
9. Acetic acid is present in ……………… while ……………… is present in lemon.
10. Excessive use of chemical fertilisers make the soil ……………… .
11. Hydrochloric acid + Sodium hydroxide → ……………… + Water
12. Acids turn blue litmus ……………… .
Answers:
1. sour, acids
2. natural
3. pink
4. neutralise, salt
5. indicator
6. indicators
7. salt
8. neutralisation
9. vinegar, citric acid
10. acidic
11. Sodium chloride
12. redTrue/False
True/False
1. All substances are either acidic or basic. [NCERT Exemplar]
2. A compound if acidic will turn all indicators red.
3. Lime water turns red litmus blue. [NCERT Exemplar]
4. Common salt dissolved in water turns blue litmus red.
5. Phenolphthalein is a natural indicator.
6. Calamine can be used to treat ant’s sting.
7. Lemon water is basic in nature.
Answers:
1. False, substances can be neutral as well.
2. False, acids do not turn all indicators red.
3. True
4. False, it does not change the colour of litmus at all.
5. False, it is a man-made indicator.
6. True
7. False, it is acidic in nature.
Match the Columns
Question 1.
Match the substances in Column I with those in Column II.
| Column 1 | Column II |
| (a Tartaric acid | (i) Soap |
| (b) Calcium hydroxide | (ii) Curd |
| (c) Formic acid | (iii) Unripe mangoes |
| (d) Sodium hydroxide | (iv) Ant’s sting |
| (e) Lactic acid | (v) Lime water |
Question 2.
Match the items in Column 1 with Column II.
| Column 1 | Column II |
| (a) Vinegar | (i) is used as indicator |
| (b) Sodium chloride | (ii) is sour to taste |
| (c) Milk of magnesia | (iii) major salt of sea |
| (d) Turmeric | (iv) changes red litmus blue |
Answers:
1.
(a)-(iii)
(b)-(v)
(c)-(iv)
(d)-(i)
(e)-(ii)
2.
(a)-(ii)
(b)-(iii)
(c)-(iv)
(d)-(i)