Haloalkanes And Haloarenes Class 12 NCERT Solutions – INTEXT Questions
Question 1. ( Haloalkanes and Haloarenes )
Write the structures of the following compounds :
(i) 2-Chloro-3-methylpentane
(ii) 1-Chloro-4-ethylcydohexane
(iii) 4-terf-Butyl-3-iodoheptane
(iv) 1, 4-Dibromobut-2-ene
(v) 1 -Bromo-4-sec-butyl-2-methylbenzene
Solution:

Haloalkanes And Haloarenes Class 12 NCERT Solutions
Question 2.
Why is sulphuric acid not used during the reaction of alcohols with Kl ?
Solution:
H2SO4 is a strong oxidising agent. Therefore, when it is used in presence of KI, it tends to convert KI to HI and finally oxidises it to I2.

Question 3.
Write structures of different dihalogen derivatives of propane.
Solution:
The structures of all possible dihalogen derivatives of propane are
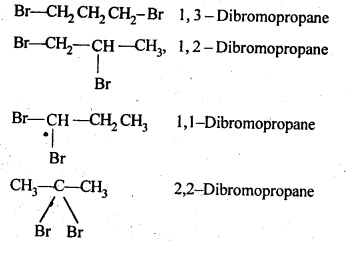
Haloalkanes And Haloarenes Class 12 NCERT Solutions Question 4.
Among the isomeric alkanes of molecular formula C5H12, identify the one that on photochemical chlorination yields
(i) A single monochloride
(ii) Three isomeric monochlorides
(iii) Four isomeric monochlorides.
Solution:
An alkane with molecular formula C5H12 can exist in the following isomeric forms :

Question 6. ( Haloalkanes and Haloarenes )
Arrange each set of compounds in order of increasing boiling points.
(i) Bromomethane, bromoform, chloromethane, dibromomethane.
(ii) 1 – Chloropropane, isopropyl chloride, 1 – chlorobutane.
Solution:
The boiling points of organic compounds depend on the strength of the intermolecular forces in them. These forces are :
(a) van der Waals forces and
(b) dipole-dipole interactions These forces are dependent on the
(i) molecular mass and
(ii) surface area of the molecules
(i) As the molecular mass of the compound increases, the boiling point also increases. Therefore the correct order is
chloromethane < bromomethane < dibromomethane < bromoform
(ii) Amongst molecules with same mass, it is the size of the molecule that determines the boiling point. Branched compounds are more compact and therefore have less surface area as compared to their straight chain counterparts and therefore lower boiling point. The order of boiling point is
iso-propyl chloride < 1-chloropropane < 1-chlorobutane
Question 7. ( Haloalkanes and Haloarenes )
Which alkyl halide from the following pairs would you expect to react more rapidly by an SN2 mechanism? Explain your answer.
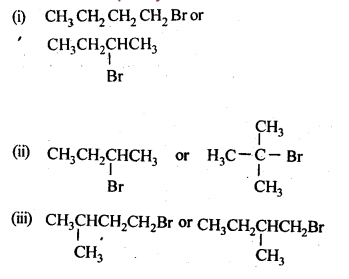
Haloalkanes And Haloarenes Class 12 NCERT Solutions:

Question 8. ( Haloalkanes and Haloarenes )
In the following pairs of halogen compounds, which compound undergoes faster SN1 reaction?
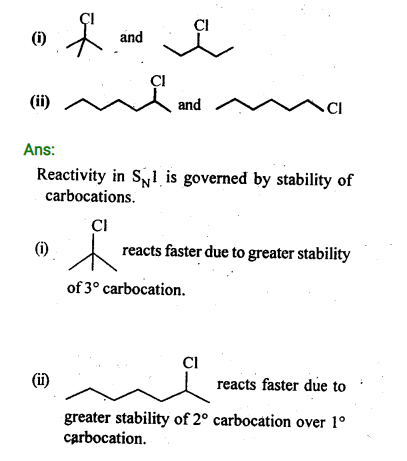
Ans

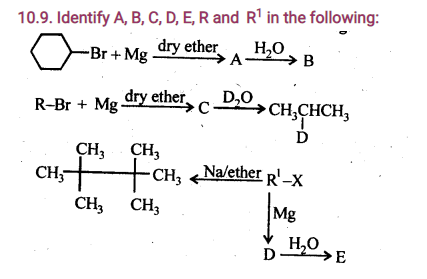
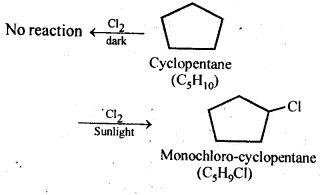
10.10. A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
Ans: The hydrocarbon with molecular formula C5H10 can either a cycloalkane or an alkene. Since the compound does not react with Cl2 in the dark, therefore it cannot be an alkene but must be a cycloalkane. Since the cycloalkane reacts with Cl2 in the presence of bright sunlight to give a single monochloro compound, C5H9Cl, therefore, all the ten hydrogen atoms of the cycloalkanes must be equivalent. Thus, the cycloalkane is cyclopentane.
NCERT EXERCISES
10.1. Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl, or aryl halides:
(i)(CH3)2CHCH(Cl)CH3
(ii) CH3CH2CH(CH3)CH(C2H5)CI
(iii) CH3CH2C(CH3)2CH2I
(iv)(CH3)3CCH2CH(Br)C6H5
(v)CH3CH(CH3)CH(Br)CH3
(vi)CH3C(C2H5)2CH2Br
(vii)CH3C(Cl)(C2H5)CH2CH3
(viii)CH3CH=C(CI)CH2CH(CH3)2
(ix)CH3CH=CHC(Br)(CH3)2
(x)P-CIC6H4CH2CH(CH3)2
(xi)m-ClCH2C6H4CH2C(CH3)3
(xii)o-Br -C6H4CH (CH3)CH2CH3
Ans: (i) 2-Chloro-3methylbutane, 2° alkyl halide
(ii) 3-Chloro-4methyl hexane, 2° alkyl halide
(iii) 1 -Iodo-2,2-dimethylbutane, 1 ° alkyl halide
(iv) l-Bromo-3, 3-dimethyl -1-phenylbutane, 2° benzylic halide
(v) 2-Bromo-3-methylbutane, 2° alkyl halide
(vi) 1-Bromo-2-ethyI-2-methylbutane, 1° alkyl halide
(vii)3-Chloro-3-methylpentane, 3° alkyl halide
(viii) 3-Chloro-5-methylhex-2-ene, vinylic halide
(ix)4-Bromo-4-methylpent-2-ene, allylic halide
(x)1-Chloro-4-(2-methylpropyl) benzene, aryl halide
(xi)1-Chloromethyl-3- (2,2-dimethylpropyl) benzene, 1 ° benzylic halide.
(xii)1-Bromo-2-(l-methylpropyl) benzene,aryl halide.
10.2. Give the IUPAC names of the following compounds:
(i) CH3CH(CI)CH (Br)CH3 (ii) CHF2CBrCIF (iii) CICH2C=CCH2Br (iv) (CCl3)3CCl
(v)CH3C(p-ClC6H4)2CH(Br)CH3 (vi)(CH3)3CCH=C(CI)C6H4I -p
Ans: (i) 2-Bromo-3-chlorobutane
(ii) 1 JBromo-1 -chloro-1,2,2-trifluoroethane
(iii) l-Bromo-4-chlorobut-2-yne
(iv)2-(Trichloromethyl)-l, 1,1,2,3,3,3- heptachloropropane
(v)2-Bromo-3,3-bis-(4-chlorophenyl) butane
(vi)l-Chloro-l-(4-iodophenyl)-3,3- dimethylbut-l-ene.
10.3. Write the structures of the following organic halogen compounds:
(i)2-ChIoro-3-methylpentane
(ii)p-Bromochlorobenzene
(iii)l-Chloro-4-ethylcyclohexane
(iv)2r (2-Chlorophenyl) -1- iodooctane
(v)2-Bromobutane
(vi)4-tert-Butyl-3-iodoheptane
(vii)1-Bromo-4-sec-butyl-2-methylbenzene
(viii)1,4-Dibromobut-2-ene
Ans:


CCl4 being symmetrical has zero dipole moment. In CHCl3, the resultant of two C – Cl dipole moments is opposed by the resultant of C – H and C – Cl bonds. Since the dipole moment of latter resultant is expected to be smaller than the former, CHCl3 has a finite dipole (1.03 D) moment.
In CH2CI2, the resultant of two C – Cl dipole moments is reinforced by resultant of two C – H dipoles, therefore, CH2CI2 (1 .62 D) has a dipole moment higher than that of CHCl3. Thus, CH2CI2 has highest dipole moment.
10.5. A hydrocarbon C5H10 does not react with chlorine in dark but gives a single monochloro compound C5H9CI in bright sunlight. Identify the hydrocarbon.
Ans: The hydrocarbon with molecular formula C5H, 0 can either a cycloalkane or an alkene.
Since the compound does not react with Cl2 in the dark, therefore it cannot be an alkene but must be a cycloalkane. Since the cycloalkane reacts with Cl2 in the presence of bright sunlight to give a single monochloro compound, C5H9Cl, therefore, all the ten hydrogen atoms of the cycloalkanes must be equivalent. Thus, the cycloalkane is cyclopentane.

10.6. Write the isomers of the compound having formula C4H9Br.
Ans:



10.10. Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene:
(I) 1-Bromo-l-methylcyclohexane
(ii) 2-Chloro-2-methylbutane.
(iii) 2,2,3-Trimethyl-3-bromopentane.
Ans:

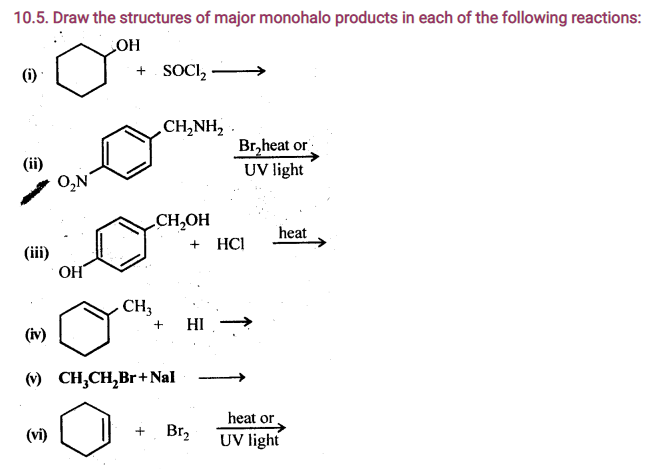
10.11. How will you bring about the following conversions?
(I) Ethanol to but-l-yne.
(ii) Ethane to bromoethene
(iii) Propene to 1-nitropropane
(iv) Toluene to benzyl alcohol
(v) Propene to propyne
(vi) Ethanol to ethyl fluoride
(vii) Bromomethane to propanone
(viii) But-l-ene to but-2-ene
(ix) 1-Chlorobutane to n-octane
(x) Benzene to biphenyl
Ans.



10.12. Explain why
(i) the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride?
(ii) alkyl halides, though polar, are immiscible with water?
(iii) Grignard reagents should be prepared under anhydrous conditions?
Ans: (i) sp2-hybrid carbon in chlorobenzene is more electronegative than a sp3-hybrid carbon in cyclohexylchloride, due to greater s-character. Thus, C atom of chlorobenzene has less tendency to release electrons to Cl than carbon atom of cyclohexylchloride.
As a result, C – Cl bond in chlorobenzene is less polar than in cyclohexylchloride. Further, due to delocalization of lone pairs of electrons of the Cl atom over the benzene ring, C-Cl bond in chlorobenzene acquires some double bond character while the C – Cl in cyclohexy! chloride is a pure single bond. In other words, C-Cl bond in chlorobenzene is shorter than in cyclohexyl chloride.
Since dipole moment is a product of charge and distance, therefore, chlorobenzene has lower dipole moment than cyclohexylchloride due to lower magnitude of negative charge on the Cl atom and shorter C-Cl distance.

10.13. Give the uses of freon 12, DDT, carbon tetrachloride, and iodoform.
Ans: Iodoform: It was earlier used as an antiseptic but the antiseptic properties are due to the liberation of free iodine and not due to iodoform itself. Due to its objectionable smell, it has been replaced by other formulations containing iodine.
Carbon tetrachloride:
Uses:
(i)As an industrial solvent for oil, fats, resins etc.and also in dry cleaning.
(ii)CCl4 vapours are highly non-inflammable, thus CCl4 is used as a fire extinguisher under the name pyrene.
(iii)Used in the manufacture of refrigerants and propellants for aerosol cans.
Freons: Freon-12 (CCl2F2) is most common freons in industrial use.
Uses: For aerosol propellants, refrigeration, and air conditioning purposes.
DDT (p -p’ – Dichloro diphenyl – trichloro ethane):
(i)The use of DDT increased enormously on a worldwide basis after World War II, primarily because of its effectiveness against the mosquitoes that spreads malaria and other insects which damages crops.
(ii) However, problems related to extensive use of DDT began to appear in the late 1940 s. Many species of insects developed resistance to DDT, it was also discovered to have a high toxicity towards fishes. DDT is not metabolised very rapidly by animals, instead, it is deposited and stored in the fatty tissues. If the ingestion continues at a steady rate, DDT builds up within the animal’s overtime.
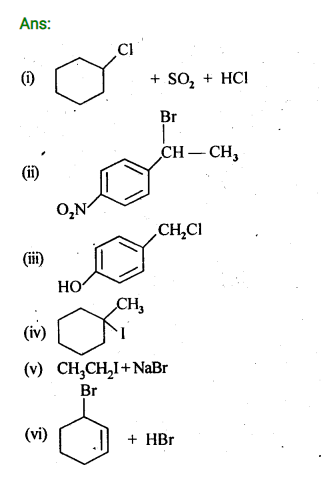
10.14. Write the structure of the major organic product in each of the following reactions:

Ans:


10.16. Arrange the compounds of each set in order of reactivity towards SN2 displacement:
(i) 2-Bromo-2-methyibutane, 1-Bromopentane, 2-Bromopentane.
(ii) l-Bromo-3-methyIbutane, 2-Bromo-2-methylbutane, 3-Bromo-2-methylbutane.
(iii) 1-Bromobutane, l-Bromo-2,2-dimethylpropane, l-Bromo-2-methylbutane, l-Bromo-3-methyl butane.
Ans: The SN2 reactions reactivity depends upon steric hindrance. More the steric hindrance slower the reaction.Thus the order of reactivity will be 1°> 2° >3°

Since in case of 1° alkyl halides steric hindrance increases in the order) n-alkyl halides, alkyl halides with a substituent at any position other than the β-position, one substituent at the β-position, two substituents at the β-position, therefore, the reactivity decreases in the same order. Thus, the reactivity of the given alkyl bromides decreases in the order:
1-Bromobutane > l-Bromo-3-methylbutane > l-Bromo-2-methyjbutane> 1-Bromo-2,2-dimethyl propane.
10.17. Out of C6H5CH2Cl and C6H5CHCIC6H5which is more easily hydrolysed by aqueous KOH.
Ans: C6H5CH2Cl is 10 aryl halide while C6H5CH(CI)C6H5 is a 2° aryl halide. In SN1 reactions, the reactivity depends upon the stability of carbocations.


10.19. How the following conversions can be carried out:
(i) Propene to propan-l-ol (ii) Ethanol to but-l-yne
(iii) l-Bromopropane to 2-bromopropane (iv) Toluene to benzyl alcohol
(v)Benzene to 4-bromonitrobenzene (vi) Benzyl alcohol to 2-phenylethanoic acid
(vii)Ethanol to propanenitrile (viii) Aniline to chlorobenzene
(ix)2-Chlorobutane to 3,4-dimethylhexane (x) 2-Methyl-1 -propene to 2-chk>ro-2-methylpropane.
(xi)Ethyl chloride to propanoic acid (xii) But-1-ene to n-butyliodide
(xiii)2-Chlropropane to 1-propanol (xiv) Isopropyl alcohol to iodoform
(xv)Chlorobenzene to p-nitrophenol (xvi) 2-Bromopropane to 1-bromopropane
(xvii)Chloroethane to butane , (xviii) Benzene to diphenyl
(xix) tert-Butyl bromide to isobutyl bromide (xx) Aniline to phenylisocyanide
Ans:




10.20. The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in presence of alcoholic KOH, alkenes are major products. Explain. (Pb. Board 2009, Haryana Board 2013)
Answer:
In aqueous medium i.e., water, KOH will be completely dissociated to give OH– ions. They being a strong nucleophile, will bring about the substitution of alkyl halides to form alcohols. At the same time, the OH” ions will be highly hydrated also. They will not be able to abstract a proton (H+) from the p-carbon atom to form alkenes. In other words, in aqueous medium, OH– ions will behave as weak base and elimination leading to alkenes will not be feasible.
In alcoholic KOH, the solution will also contain ethoxide ions (C2H5O–) in addition to OH– ions. They being a stronger base than OH– ions, will abstract a H+ ion from the β-carbon atom giving alkene as the product as a result of dehydrohalogenation.
10.21. Primary alkyl halide C4H9Br (a) reacted with alcoholic KOH to give compound (b) Compound (b) is reacted, with HBr to give (c) which is an isomer of (a). When (a) is reacted with sodium metal it give compound (d), C8H18 which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions.
Ans: (i) There are two primary alkyl halides having the molecular formula, C4H9Br.


10.22. What happens when .
(i) n-butyi chloride is treated with alcoholic KOH.
(ii) bromobenzene is treated with Mg in the presence of dry ether.
(iii) chlorobenzene is subjected to hydrolysis.
(iv) ethyl chloride is treated with aqueous. KOH.
(v) methyl bromide is treated with sodium in the presence of dry ether,
(vi) methyl chloride is treated with KCN.
