


- This is because it is property of solution that polar solutes dissolve in polar solvents like Water, ethanol, formaldehyde and chloroform whereas non polar solutes in nonpolar solvents like pyridine, toluene, and hexane. This phenomenon can be easily defined as like dissolves like.
- The phenomenon of increase in the concentration of solution due to dissolving of solid solute to the solvent is known as dissolution.
- The phenomenon of collision of solute particles in a solution resulting in the separation of the solute particles from the solution is known as crystallization.
- A phase when number of solute particles going into solution is equal to the solute particles separating out of the solution is known as equilibrium. The concentration of solute remains constant at this stage.
- A solution in which no more solute can be dissolved into the solvent at the same temperature and pressure is known as saturated solution.
- Whereas a solution in which more solute can be dissolved in the solvent at the same temperature and pressure is known as unsaturated solution.
- According to Le Chateliers Principle in a saturated solution, if the dissolution process is endothermic (Δsol H > 0), the solubility should increase with rise in temperature but if the dissolution process is exothermic (Δsol H > 0) the solubility should decrease.
- Pressure does not have crucial effect on solubility of solids in liquids because they remain unaffected to pressure due to the high incompressibility of solids and liquids.

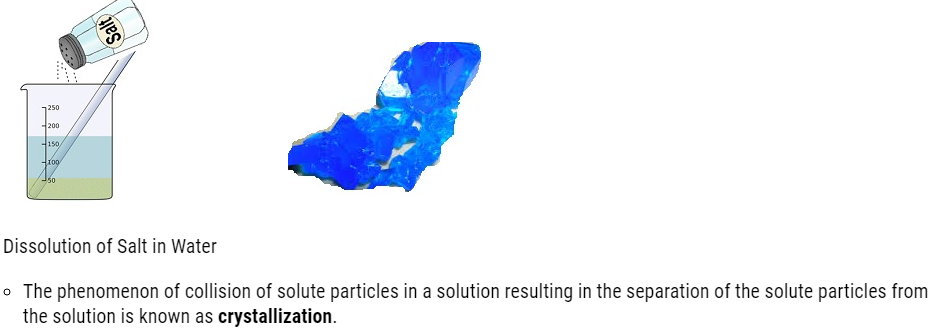
Crystal of copper Sulfate obtained by the crystallization of Copper Sulfate solution
- A phase when number of solute particles going into solution is equal to the solute particles separating out of the solution is known as equilibrium. The concentration of solute remains constant at this stage of dynamic equilibrium.
- Solubility of gas in liquid is highly affected by temperature and pressure.
- Increase in temperature reduces the solubility of gases in liquids because on dissolving gas molecules in liquid the process of dissolution is similar to condensation leading to the evolution of heat. Therefore, dissolution is an exothermic process, due to which the solubility decreases with increase of temperature.
- Increase in pressure by compressing the gas to a smaller volume increase the number of gaseous particles per unit volume over the solution and also the rate at which the gaseous particles are striking the surface of solution to enter it leading to the increase in the solubility of gases.
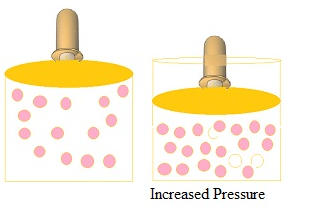
Henry’s law
- Henry’s Law establishes aquantitative relation betweenpressure and solubility of a gasin a solvent.
- This law is for gas-liquid solution.
- According to the law at a constant temperature, thesolubility of a gas in a liquid is directly proportional to thepressure of the gas.
- It can also be stated as – The partial pressure of the gas in vapour phase (p) is proportional to the mole fraction of the gas (x) in the solution.
- Mathematically,
p = KH x where KH = Henry’s law constant.
- At same temperature different gasses have different KH
- It is a function of the nature of the gas. At a given pressure increasing value of KH implies lower solubility of the gas in the liquid.
- Value of KH increases with the increase in temperature therefore solubility of gases increases with decreasing temperature. Due to this reason cold water is more sustainable for aquatic life than warm water.
Problem: H2S, a toxic gas with rotten egg like smell, is used for the qualitative analysis. If the solubility of H2S in water at STP is 0.195 m, calculate Henry’s law constant.
Solution:
It is given that the solubility of H2S in water at STP is 0.195 m, i.e., 0.195 mol of H2S is dissolved in 1000 g of water.
Moles of water = 1000g / 18g mol-1 = 55.56 mol
∴Mole fraction of H2S, x = Moles of H2S / Moles of H2S+Moles of water 0.195 / (0.195+55.56)= 0.0035
At STP, pressure (p) = 0.987 bar
According to Henry’s law:
p = KH x ⇒ KH = p / x
= 0.0987 / 0.0035 bar
= 282 bar


Short Notes on Solubility & Henry Law:-


2.12. State Henry’s law and mention some of its important applications. (NCERT Book)
Sol:
Henry’s law: The solubility of a gas in a liquid at a particular temperature is directly proportional to the pressure of the gas in equilibrium with the liquid at that temperature.
or
The partial pressure of a gas in vapour phase is proportional to the mole fraction of the gas (x) in the solution. p = KHX
where KH is Henry’s law constant.
Applications of Henry’s law :
(i) In order to increase the solubility of CO2 gas in soft drinks and soda water, the bottles are normally sealed under high pressure. Increase in pressure increases the solubility of a gas in a solvent according to Henry’s Law. If the bottle is opened by removing the stopper or seal, the pressure on the surface of the gas will suddenly decrease. This will cause a decrease in the solubility of the gas in the liquid i.e. water. As a result, it will rush out of the bottle producing a hissing noise or with a fiz.
(ii) As pointed above, oxygen to be used by deep sea divers is generally diluted with helium inorder to reduce or minimise the painfril effects during decompression.
(iii) As the partial pressure of oxygen in air is high, in lungs it combines with haemoglobin to form oxyhaemoglobin. In tissues, the partial pressure of oxygen is comparatively low. Therefore, oxyhaemoglobin releases oxygen in order to carry out cellular activities.
2.35. Henry’s law constant for the molality of methane in benzene at 298 K is 4.27 x 105 mm Hg. Calculate the solubility of methane in benzene at 298 K under 760 mm Hg.(NCERT Book)
Solution:
mm Hg (at 298 K)