Reynolds Number
- Reynolds number is a dimensionless number, whose value gives an idea whether the flow would be turbulent or laminar.
- Types of flow are classified as 2 types:laminar flow and turbulent flow.
- Reynolds number helps us to determine whether the flow is laminar or turbulent.
- It is denoted by Re. where ‘e’ shows Reynolds.
- Expression: Re=ρvd/ η;
- where ρ = density of the fluid,
- v=velocity of the fluid,
- d=diameter of the pipe through which the fluid flows
- η=viscosity of the fluid.
How does Reynolds number (Re) distinguish laminar flow from tubular?
- If the value of Reynold’s number (Re) reaches 1000 then the flow is laminar.
- When the value of Reynold’s number(Re)is greater than 2000 then the flow is turbulent.
- If the value of (Re)is between 1000 and 2000 then the flow is unstable.The flow is in intermediate stage.
- At this state it has some characteristics of laminar flow and some of turbulent flow.

Alternative expression of Re: Inertial force/force of viscosity
- By using Re= ρvd/ η
- Multiplying both numerator and den by v:- Re= ρv2d/ ηv
- By rearranging, ρv2/( ηv/d)
- Multiplying both numerator and den by A:- Re= ρv2A/(ηv/d)A
- Where
- ρv2A = Inertial force
- (ηv/d)A = Force of viscosity
- Where
(a)Calculating inertial force
- Inertial force = ma
- =ρV xv/t = (ρVxAxdisplacement)/t
- =ρvAv =ρv2A
(b)Calculating Force of viscosity:-
- Coefficient of viscosity η =stress/shearing strain
- F/A/(x/lt)
- F/A/v/l =Fl/Av
- η=Fl/Av
- F = ηAv/l
- =(ηv/l)A (Expression is same as )
Turbulence:boon or bane
- Turbulence has both advantages and disadvantages.
- Advantages:-
- Promotes mixing and increases the rates of transfer of mass,momentum and energy. For example: – Mixer and Grinder or a juice mixer.

Grinding of flour
- Disadvantages:-
- Dissipates Kinetic energy in the form of heat.
Problem:- The flow rate of water froma tap of diameter 1.25 cm is 0.48 L/min.The coefficient of viscosity of water is10-3 Pa s. After sometime the flow rate isincreased to 3 L/min. characterise the flowfor both the flow rates.
Answer:- Let the speed of the flow be v and thediameter of the tap be d = 1.25 cm. Thevolume of the water flowing out per second isQ = v × π d2 / 4
v = 4 Q / d2
We then estimate the Reynolds number to be
Re = 4 ρ Q / π d η
= 4 ×103 kg m–3 × Q/(3.14 ×1.25 ×10-2 m ×10-3 Pa s)
= 1.019 × 108 m–3 s Q
Since initially
Q = 0.48 L / min = 8 cm3 / s = 8 × 10-6 m3 s-1,
We obtain,
Re = 815
Since this is below 1000, the flow is steady.After some time when
Q = 3 L / min = 50 cm3 / s = 5 × 10-5 m3 s-1,
We obtain,
Re = 5095
Liquid Surfaces
- Certain properties of free surfaces:-
- Whenever liquids are poured in any container they take the shape of that container in which they are poured and they acquire a free surface.
- Consider a case if we pour water inside the glass it takes the shape of the glass with a free surface at the top.
- Top surface of the glass is a free surface. Water is not in contact with anything else,it is in contact with the air only.
- This is known as free surfaces.
- Liquids have free surfaces. As liquids don’t have fixed shape they have only fixed volume.
- Free surfaces have additional energy as compared to inner surfaces of the liquid.

Surface Energy
- Surface energy is the excess energy exhibited by the liquid molecules on the surface compared to those inside the liquid.
- This means liquid molecules at the surface have greater energy as compared to molecules inside it.
- Suppose there is a tumbler and when we pour water in the tumbler,it takes the shape of the tumbler.
- It acquires free surface.
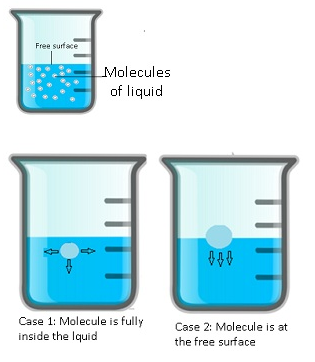
- Case 1: When molecules are inside the liquid:-
- Suppose there is a molecule inside the water,there will be several other molecules that will attract that molecule in all the directions.
- As a result this attraction will bind all the molecules together.
- This results in negative potential energy of the molecule as it binds the molecule.
- To separate this molecule huge amount of energy is required to overcome potential energy.
- Some external energy is required to move this molecule and it should be greater than the potential energy.
- Therefore in order to separate this molecule a huge amount of energy is required.
- Therefore a large amount of energy is required by the molecules which are inside the liquid.
- Case2: When the molecules are at the surface:-
- When the molecule is at the surface, half of it will be inside and half of it is exposed to the atmosphere.
- For the lower half of the molecule it will be attracted by the other molecules inside the liquid.
- But the upper half is free. The negative potential energy is only because of lower half.
- But the magnitude is half as compared to the potential energy of the molecule which is fully inside the liquid.
- So the molecule has some excess energy, because of this additional energy which the molecules have at the surface different phenomenon happen like surface energy, surface tension.
- Liquids always tend to have least surface are when left to itself.
- As more surface area will require more energy as a result liquids tend to have least surface area.
Surface energy for two fluids in contact
- Whenever there are two fluids,in contact, surface energy depends on materials of the surfaces in contact.
- Surface energy decreases if the molecules of the two fluids attract.
- Surface energy increases if molecules of the two fluids repel.