Matter
Anything that has mass and occupies space is defined as matter. For example:-soap, notebook, pencil, apple, battery etc.
Matter can exist in 3 physical states namely solid, liquid and gas. (Physical Classification)
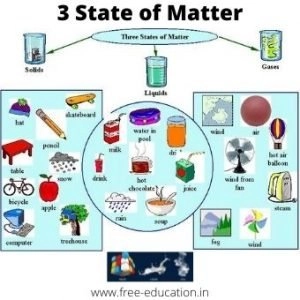
Solids
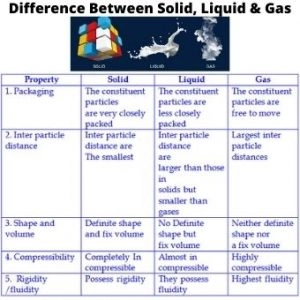
- In case of solids, the constituent particles are held very close to each other in an orderly fashion and there is not much freedom of movement.
- Solids have definite shape and definite volume.
- For example: – Apple it has definite shape as well as definite volume.
Liquids
- In liquids, the particles are close to each other but they can move around.
- Liquids have definite volume but not definite shape. They take the shape of the container in which they are placed.
- For example: Water it takes the shape of the tumbler in which it is poured but has volume.
Gases
- In gases, the particles are far apart as compared to those present in solid or liquid states and their movement is easy and fast.
- If container in which they are placed.
- For example: – Smoke does not have definite shape or volume.
Note:-
- All the 3 states of matter are inconvertible among each other.
- Consider solid if we melt it , it changes into liquid and if it is further heated it becomes
- When the gas is condensed they change to liquid. And when the liquid is freezed it becomes solid.
- By sublimation solid changes into gas and gas by deposition it becomes
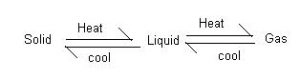
Matter can be further classified as
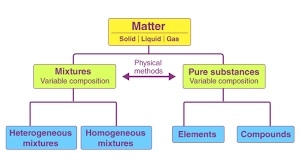
Pure Substances
Pure substances have fixed composition. For example: – Copper, silver, gold, water, glucose are some examples of pure substances.
-
- Glucose contains carbon, hydrogen and oxygen in a fixed ratio and thus, like all other pure substances has a fixed composition.
- Composition of carbon, hydrogen and oxygen in sugar will be always C12H22O11.They will be always in fixed ratio.
- Also, the constituents of pure substances cannot be separated by simple physical methods.
- Pure substances are further classified into elements and compounds.
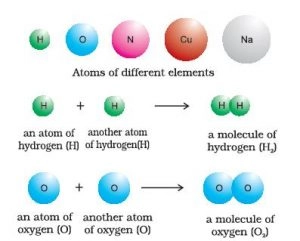
Elements:-
An element consists of only one type of particles. These particles may be atoms or molecules.
- For example: – Sodium (Na), copper (Cu), silver (Ag), hydrogen (H), oxygen (O) atoms etc. They contain only one type of atoms.
Compounds:-
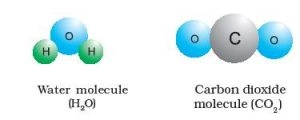
- When two or more atoms of different elements combine, the molecule of a compound is obtained.
- For example: – Water (H20), Ammonia (NH3), Sugar, carbon dioxide (CO2).
Properties of Compound:-
The properties of a compound are different from those of its constituent elements.
For example: – Hydrogen (H2) and Oxygen (O2) are gases whereas the compound formed by their combination i.e., water (H20) is a liquid.
Hydrogen burns with a pop sound and oxygen is a supporter of combustion, but water is used as a fire extinguisher.
Constituents of a compound cannot be separated into simpler substances by physical methods. They can be separated by chemical methods.
A mixture contains two or more substances present in it (in any ratio) which are called its components.
For example:-Air, sugar solution, mixture of pulse and stone.
Homogeneous or Heterogeneous
A mixture can be homogeneous or heterogeneous.
In homogeneous mixture, the components completely mix with each other and its composition is uniform throughout.
For example: – Air, Sugar solution.
In heterogeneous mixtures, the composition is not uniform throughout and sometimes the different components can be observed.
For example: – Mixture of dal mot and bhujia, badam etc.
The components of a mixture can be separated by using physical methods such as simple hand picking, filtration, crystallisation, distillation etc.
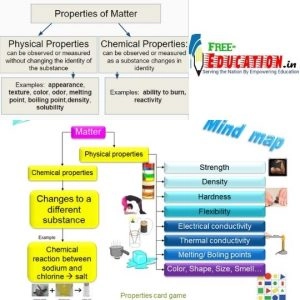
Properties of Matter
Every substance has characteristic properties. These properties can be classified into 2 categories: – Physical and Chemical properties.
Physical properties
- They are those properties which can be measured or observed without changing the identity or the composition of the substance.
- Some examples of physical properties are colour, odour, melting point, boiling point, density etc.
Chemical properties
- The chemical properties require a chemical change to occur.
- Chemical properties are characteristic reactions of different substances; these include acidity or basicity, combustibility etc.
Question of these Topics
Q. 1: Define Matter?
Q. 2: Explain physical classification of matter?(Imp.)
Q. 3: What is the difference between Homogeneous and Heterogeneous Matter.(Imp.)
Q. 4: What are the Physical Properties of Matter?(Imp.)
Q. 5: What do you understand by chemical properties of matter?(Imp.)
Q. 6: Write the three points of difference between compound and mixture?(Imp.)
Q. 7: Classify the following as pure substances or mixture –
(a) ethyl alcohol (b) oxygen (c) blood (d) carbon (e) steel (f) distilled water
Ans. Pure substance – ethyl alcohol, oxygen, carbon, distilled water
Mixture – blood, steel.