Ionization Enthalpy of alkaline earth metal
- Ionization energy is the quantity of energy required to remove loosely bound electron from an atom.
- They have low ionization enthalpies due to fairly large size of the atoms.
- Ionization enthalpy decreases with the increase in atomic size down the group.

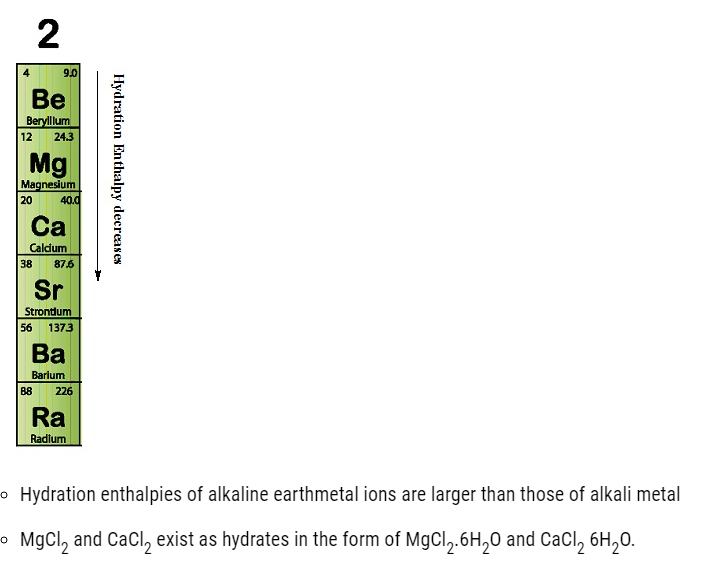
Hydration Enthalpy of alkaline earth metal
- The energy released as a result of the formation of new bonds between ions and water molecules is called hydration enthalpy.
- Hydration enthalpiesof alkaline earth metal ions decrease with the increasing ionic size down the group.
Physical Properties of alkaline earth metal
- The alkaline earth metals are silverywhite, soft and lustrous.
- But they are somewhat harder than alkali metals.
- Beryllium andmagnesium are greyish in colour.
- Melting and boiling points of these metalsare higher than corresponding alkali metal.
- They have low ionizationenthalpy which makes them strongly electropositive that increasesdown the group from Be to Ba.
- When exposed to flame calcium, strontium and barium imparts brick red, crimson and apple green colours

Chemical Properties of alkaline earth metal
(i) Reactivity towards air and water
Beryllium and magnesium are kinetically inert to oxygen and water due to an oxide film on their surface.
Powdered beryllium burns brilliantly on ignition in air to give BeO and Be3N2.
Magnesium is more electropositive and burns with dazzling brilliance in air to give MgO and Mg3N2.
Calcium, strontium and barium react with water and air.
(ii) Reactivity towards the halogens
They combine with halogen to form halides.
M+ X2 –> MX2
(iii) Reactivity towards hydrogen
Alkaline earth metals elements except beryllium combine with hydrogen upon heating to form their hydrides.
But BeH2 can be prepared by following process too.
2BeCl2 +LiAlH4 –> 2BeH2 +LiCl + AlCl3
(iv) Reactivity towards acids
They readily react with acids liberating dihydrogen.
M +2HCl –> MCl2 + H2
(vi) Solutions in liquid ammonia
Dissolve in liquid ammonia to give deep blue black solutions forming ammoniated ions.
M + (x+y) NH3 –> [M (NH3) x] 2+ + 2[e (NH3) y]–
