About Lesson
PART 1
PART 2
What is Chemistry?
- It can also be defined as that branch of science that deals with the study of the composition, properties and interaction of matter.
- Chemistry is the science of atoms, molecules and their transformations.
Branches of Chemistry
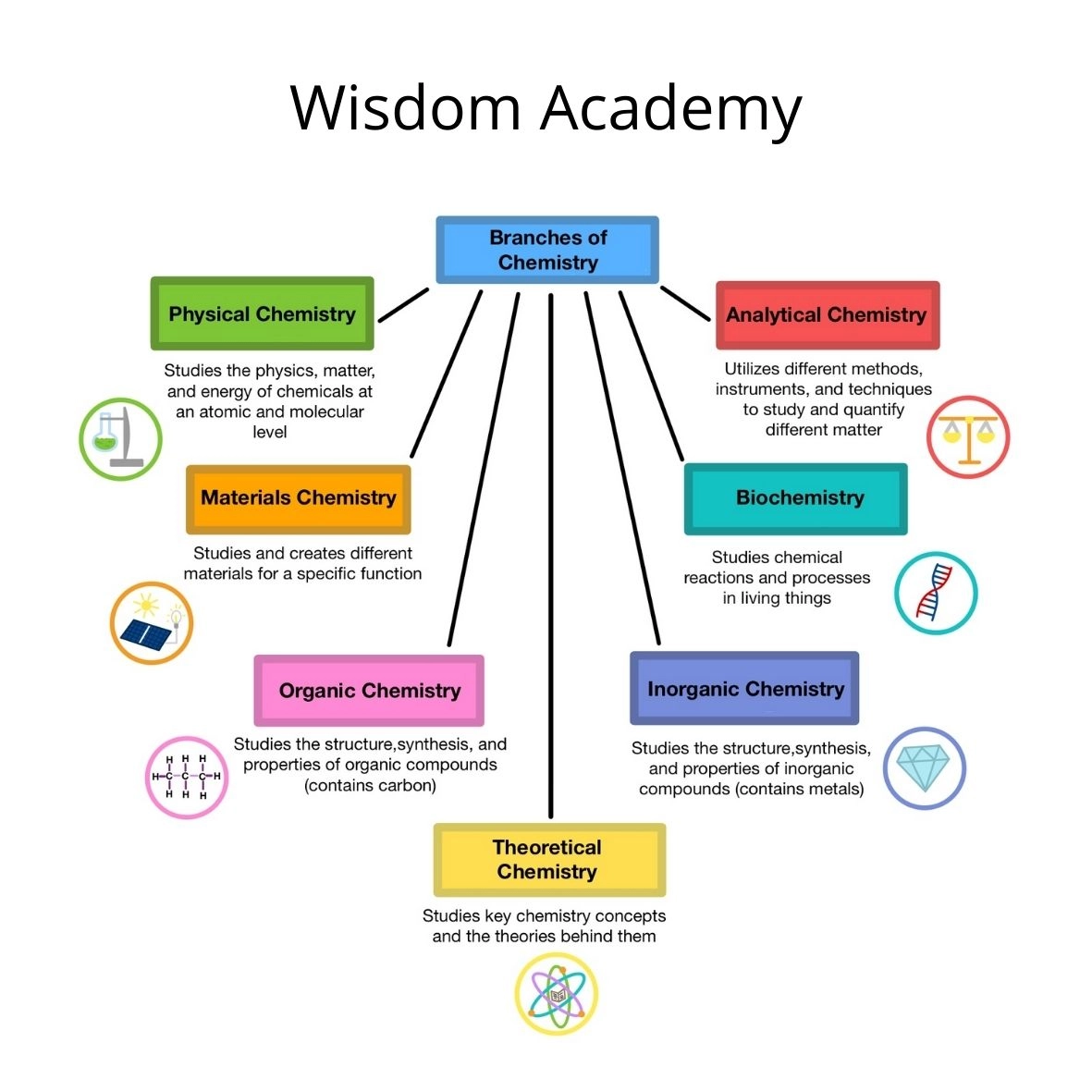
- Organic Chemistry:- This branch deals with study of carbon compounds especially hydrocarbons and their derivatives
- Inorganic Chemistry:- This branch deals with the study of compounds of all other elements except carbon. It largely concerns itself with the study of minerals founda in the Earth’s crust.
- Physical Chemistry:- The explanation of fundamental principles governing various chemical phenomena is the main concern of this branch. It is basically concerned with laws and theories of the different branches of chemistry.
- Industrial Chemistry:- The chemistry involved in industrial processes is studies under this branch.
- Analytical Chemistry:- This branch deals with the qualitative and quantitative analysis of various substances.
- Biochemistry:- This branch deals with the chemical changes going on in the bodies of living organisms; plants and animals.
- Nuclear Chemistry:- Nuclear reactions, such as nuclear fission, nuclear fusion, transmutation processes etc. are studied under this branch.
Knowledge of chemistry helps us to know:-
- The properties of different substances like diamond, coal, plastics etc.
- In preparation of soaps like liquid or powder.
- The fabrics, plastic toys, paper, pencil, eraser, sugar cubes, petrol or diesel etc.
- In the manufacture of different types of fertilizers, pesticides and medicines.

Chemistry helps us to answer the following questions:-
- To count the number of atoms and molecules in a given mass of matter?
- How to see the atoms and molecules?
Advantages of studying chemistry
- With a better understanding of chemical principles it has now become possible to design and synthesise new materials having specific magnetic, electric and optical properties.
- Safer alternatives to environmentally hazardous refrigerants like CFCs (chlorofluorocarbons), responsible for ozone depletion in the stratosphere, have been successfully synthesised.
Problems which can be tackled with the study of chemistry:-
- Management of Green House gases like methane, carbon dioxide etc.
- Understanding of bio-chemical processes, use of enzymes for large-scale production of chemicals and synthesis of new exotic materials are some of the intellectual challenges for the future generation of chemists.
- Understanding of bio-chemical processes, use of enzymes for large-scale production of chemicals.
Question may be asked on this topic.
Q.1: Define Chemistry and It’s Branch?
Q.2: How can we use chemistry in our day to day life?
Q.3: What are the advantages to learn chemisty?
Join the conversation